 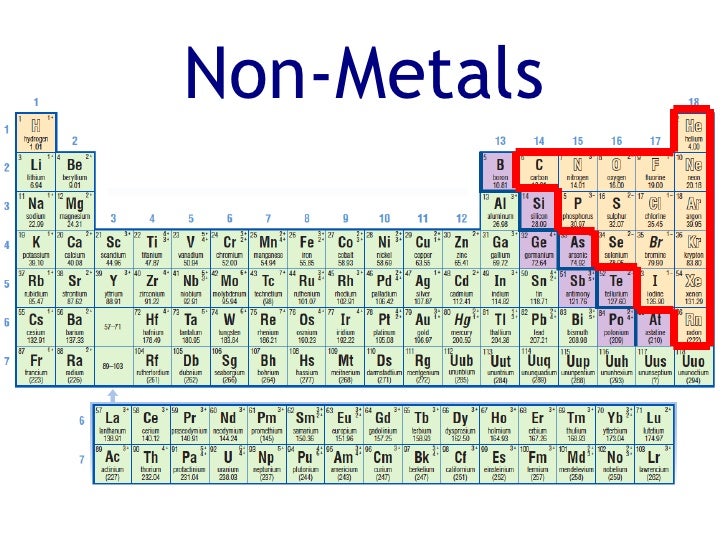
It has been known for over 120 years that metalloids have a predominately nonmetallic chemistry (Newth 1894 Friend 1914). The characteristics of the corrosive nonmetals and intermediate nonmetals are subsequently elaborated in this article, the latter at some length. This results in a balanced 6–5–6–6 distribution of the elements concerned. As the metals have been subdivided into four broad classes, so can the nonmetals be subdivided into like classes of noble gases corrosive nonmetals intermediate nonmetals and metalloids. And some or all of the metalloids may or may not be included in the applicable sections.Ī richer, complementary taxonomy is introduced in this article. Thus, in a textbook, there may be separate sections on hydrogen carbon nitrogen and phosphorus and oxygen, sulfur, and selenium. In Steudel’s Chemistry of the nonmetals (1977, preface)Īfter highlighting the halogens and the noble gases, authors have always found it difficult to treat the remaining nonmetals on a holistic rather than group-by-group basis. “The marvellous variety and infinite subtlety of the non-metallic elements, their compounds, structures and reactions, is not sufficiently acknowledged in the current teaching of chemistry.” The literature is sparse when it comes to holistic treatments of their chemistry. Wikipedia ( 2019a) records 15 different category names for these elements located between the transition metals and the metalloids, including ‘other metals’. In some respects, the frontier metals can be regarded as the forgotten metals. Being adjacent to the non-metals, their crystalline structures tend to show covalent or directional bonding effects, having generally greater complexity or fewer nearest neighbours than other metallic elements (Russell and Lee 2010, p. A minority are moderately electropositive (zinc, for example) (Kneen et al. Most of them, such as tin and bismuth, are chemically weak. The frontier metals, as a class, are adjacent to the dividing line between metals and nonmetals. A small number, such as zirconium, are more strongly electropositive several others are chemically very weak (or noble), like platinum, with these representing the noble metals. The transition metals are, for the most part, moderately to weakly electropositive in nature (Kneen et al. They are mostly strongly electropositive metals, with a few of the light actinoids (uranium to americium) being only moderately electropositive. The active metals are here considered to encompass groups 1–3, the lanthanoids and actinoids, and aluminium in group 3. Footnote 1 A fourth class, the noble metals, is a subset of the transition metals (MacKay et al. The metals can be divided into broad classes, as: active metals transition metals and frontier metals (Russell and Lee 2005, p. A few borderline elements are sometimes identified as metalloids. I hope you have found this article helpful in solving your doubts.įeel free to ask me in the comments, if you have any doubts.In general, the elements of the periodic table can be divided into metals and nonmetals. Then we discussed the total number of metals, Nonmetals and Metalloids present on the Periodic table. In the very beginning of this article, I showed you the Periodic table labeled with metals, Nonmetals and Metalloids. All these known elements are classified in three major categories based on their metallic and nonmetallic characteristics. Periodic table consists of total 118 elements. Get Info + HD Image of Periodic Table Summary You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Why are metalloids also known as semimetals? Free Gift for you: Interactive Periodic Table List of metalloids with atomic number, symbol and names of element.Ģ). I want to keep this article short and I have already explained the reason why astatine and polonium are not classed as metalloids. There are 6 commonly known Metalloids on the Periodic table.Īstatine and Polonium also shows some characteristics of Metalloids.īut according to some researchers, Astatine and Polonium are not classes as Metalloids. How many Metalloids are on the Periodic table? They are found between the metals and nonmetals.Įlements which have the metallic properties as well as nonmetallic properties are classified as Metalloids.īecause of this reason, they are also known as semimetals. Above picture shows you the labeled Metalloids on the Periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
